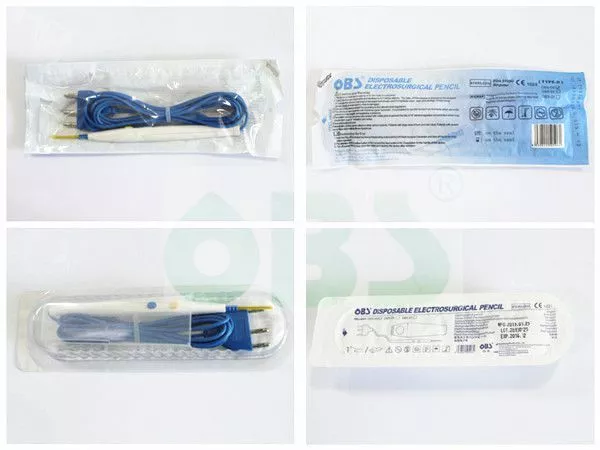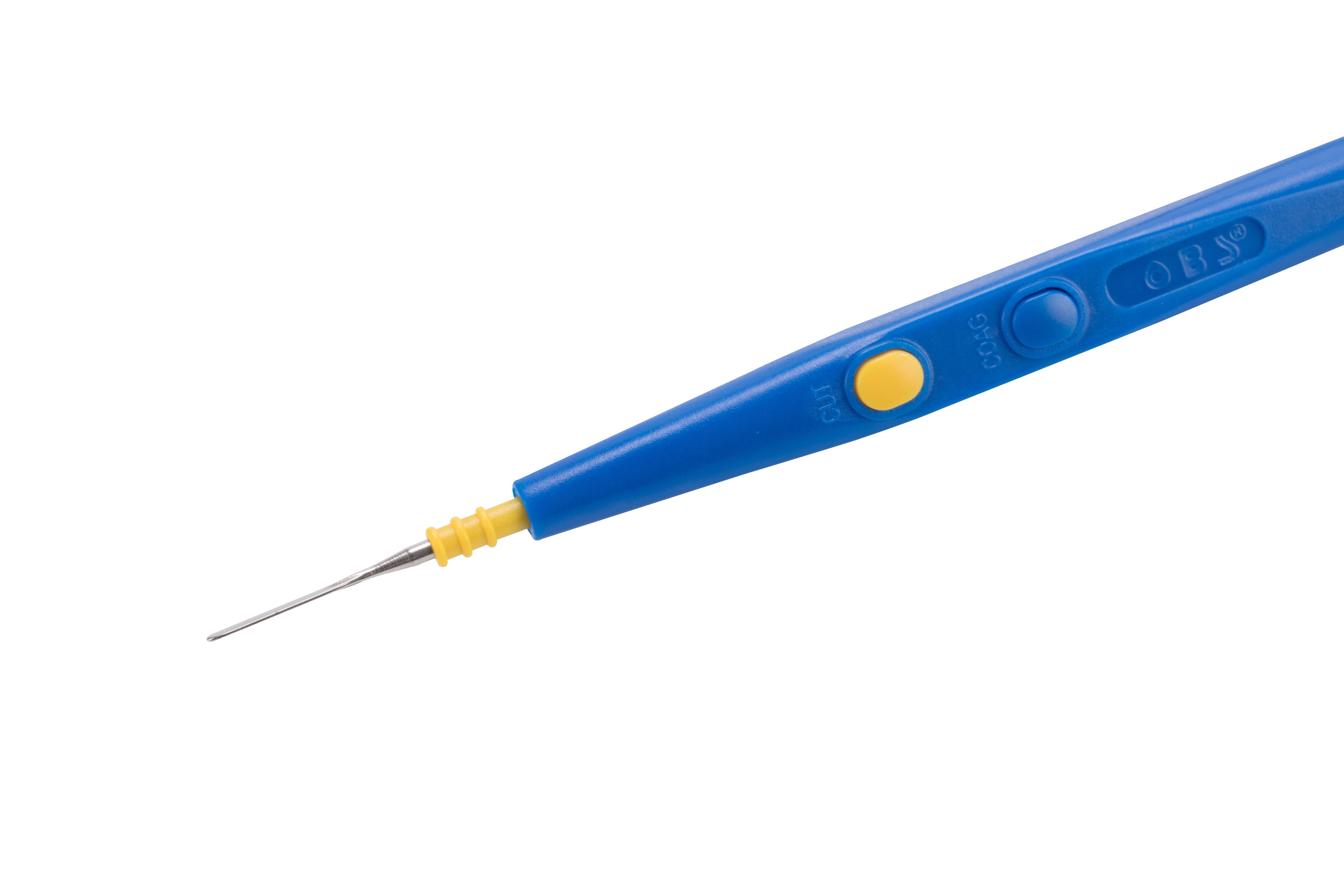

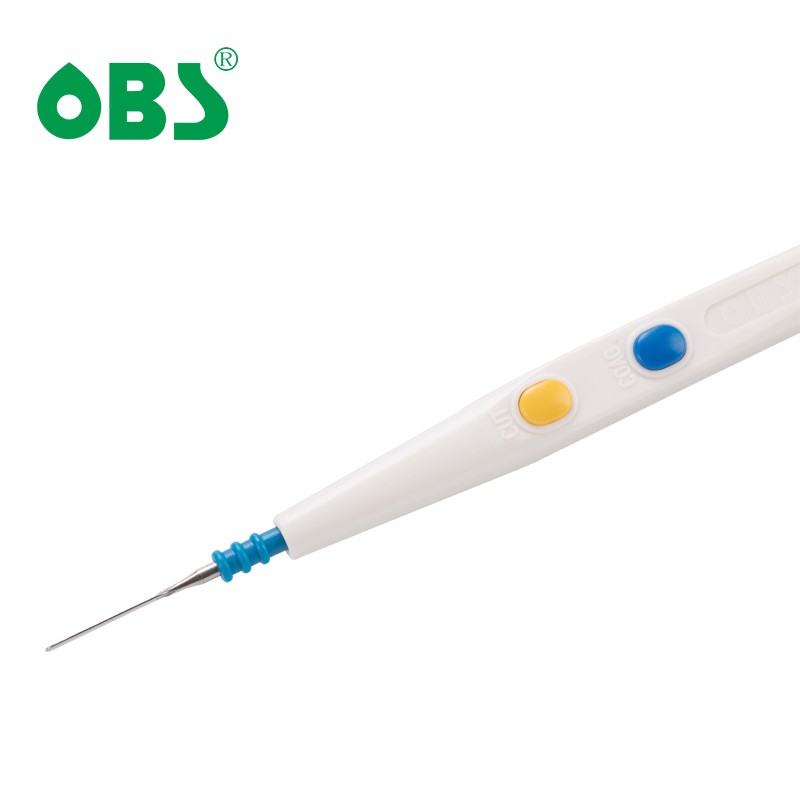
| Place of Origin: | Guangdong, China |
| Brand Name: | OBS |
| Model Number: | #0039 |
| Properties: | The Basis of Surgical Instruments |
| Type: | Knife |
| Mode: | OBS-Db #0039 |
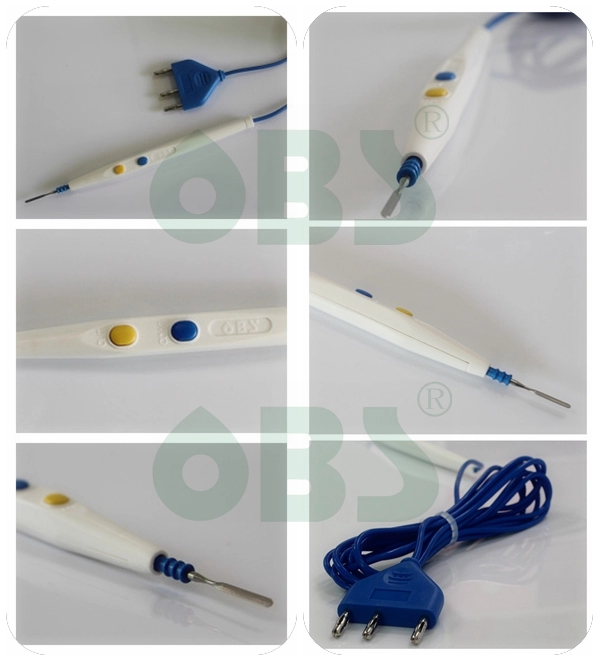
| Color: | white/blue |
| Sterilization: | EO |
| Shell Lenght: | 15.3cm |
| Cable Lenght: | 3m/5m |
| Plug: | 3-pin plug |
| Weight Per Piece: | 0.068kg |
Video Description
Disposable Electrosurgical Pencil
( Disposable ESU Pencil, Cautery Pencil )
FDA 510(k) Approval
Company FDA Registation No.: 3007748793
Device K No.:K092634
CE Mark
Range of Using:
Match with Electrosurgical Generator(Unit), Radio Frequency Generator(Unit) and other High Frequency Equipment.
Type:
D: Disposable
These devices are designed to be used as accessories in conjunction with those electrosurgical units and electrodes. These devices are single use supplied sterile. Check plug matching on generator base and connect. Press CUT/yellow button for cutting and COAG/blue button for coagulation. Maximum voltage is not be exceed 90KV peak
Product feature:
Handswitching pencils are available with either push-button or rocker switch controls to accommodate the surgeon's preference. Switches have a high level of tactile feedback - surgeons can hear and feel the switch engage, preventing accidental activation of the device.
Designed with the surgeon in mind
-Slim and lightweight, tapered ergonomic design, streamlined design,
-Slip-proof handling and water resistant Rocker or Push-Button Switch
-Switch and buttons designed for smooth activation
-Equip with a range of monopolar cutting and coagulation electrodes to satisfy all users requirements.
-With hexagonal shape to protect against inadvertent twisting, the electrodes can be locked safely into place and are easily exchanged and replaced.
Electrical Performance Data
Maximal Output Current | Frenquency Confine | Current Resistence | Output Power |
1A | 0-1.0MHz | 200Ω | 50-80W |
Warning
These devices should never be used when:
There is visible evidence of damage to the exterior of the device such as cracked or damaged plastic or connector damage.
These devices fail the inspection described herein.
In the presence of flammble gases, flammable prep solutions or drapes. Oxidizing. Gases such as Nitrous Oxide (N2O),or in oxygen-riched environments.
Do not used monopolar electrosurgery on small appendages, as in circumcision or finger surgery.
Packing
Content | Net Weight | Gross Weight | Carton Size | |
Paper Packaging | 100 pcs/ctn | 6.8kg | 7.3kg | 36*27*28 |
Blister Packaging | 25 pcs/ inner ctn; 4 inner ctns/outer ctn | 9.8kg | 11.5kg | 53.5*36*40 |